Erhalten Sie Zugang zu diesem und mehr als 300000 Büchern ab EUR 5,99 monatlich.
- Herausgeber: Quintessence Publishing Co, Inc
- Kategorie: Fachliteratur
- Sprache: Englisch
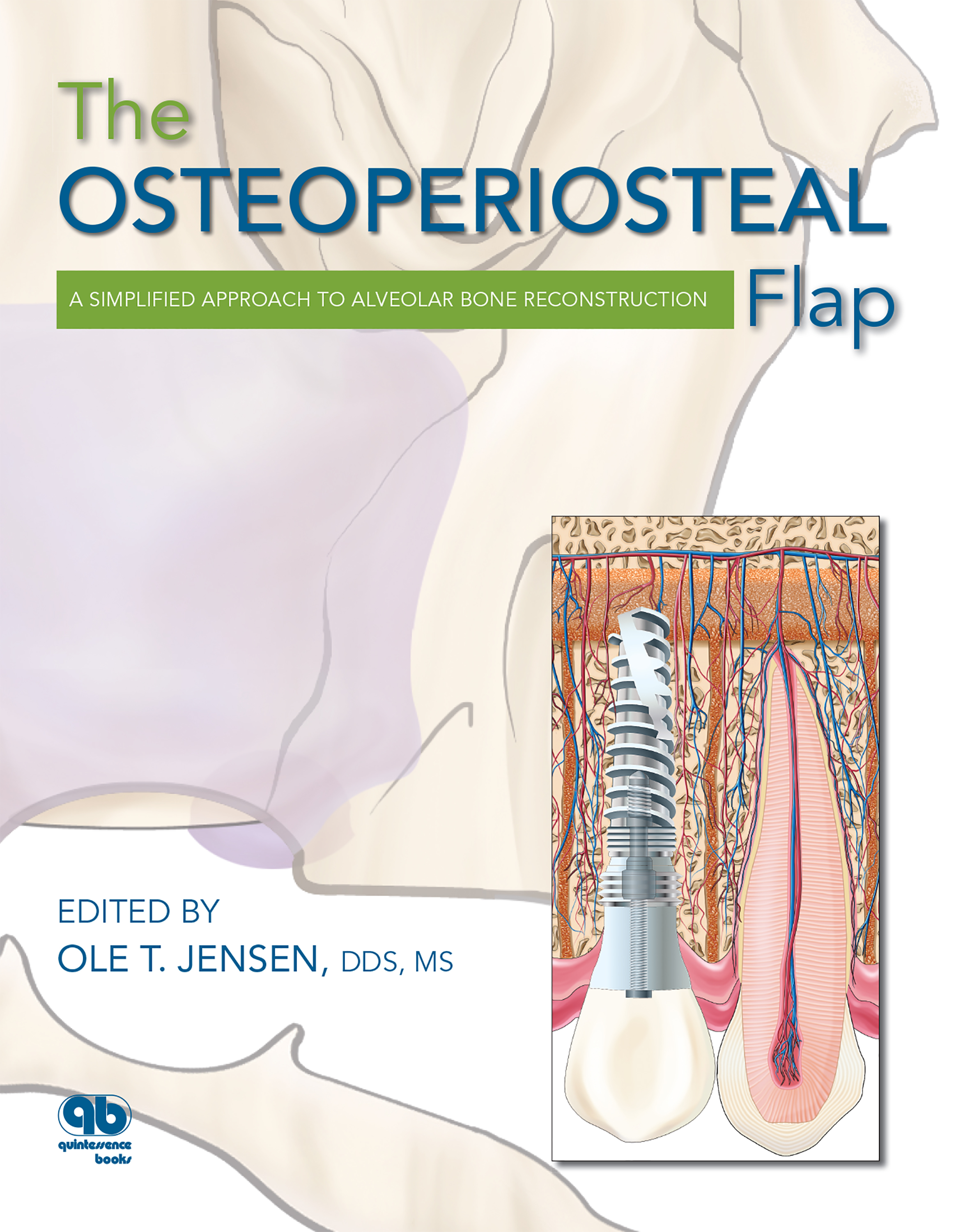
This elaborately illustrated book takes a fresh look at alveolar bone reconstruction, positing that the vitality of the gingiva-alveolus-implant complex is more important than simple implant longevity. With the use of osteoperiosteal flaps, the surgeon manipulates available bone to recover what is missing in a very specific way: endosteally. This relatively closed wound approach seems to spontaneously activate the epigenetic signal within the gingivoalveolar complex, and the augmentation develops in a manner analogous to primordial growth. Soft tissue generally follows suit, and implant therapy can commence, creating a functional gingiva-alveolus-implant matrix. Once mastered, bone flaps can almost entirely eliminate the need for block grafting or guided bone regeneration. The culmination of many years of clinical research, this volume presents procedures for various osteoperiosteal flaps. It is intended for the private practitioner who must use techniques that work consistently, minimize morbidity, and are simple and relatively quick to perform. The osteoperiosteal flap, in its various permutations, fulfills these criteria beautifully.
Sie lesen das E-Book in den Legimi-Apps auf:
Seitenzahl: 510
Veröffentlichungsjahr: 2019
Das E-Book (TTS) können Sie hören im Abo „Legimi Premium” in Legimi-Apps auf:
Ähnliche
The Osteoperiosteal Flap: A Simplified Approach to Alveolar Bone Reconstruction
Ole T. Jensen
Library of Congress Cataloging-in-Publication Data
The osteoperiosteal flap : a simplified approach to alveolar bone reconstruction / edited by Ole T. Jensen. p. ; cm. Includes bibliographical references and index.
9780867158922
1. Dental implants. 2. Alveolar process--Growth. 3. Flaps (Surgery) 4. Bone-grafting. I. Jensen, Ole T. [DNLM: 1. Alveolar Ridge Augmentation--methods. 2. Bone Regeneration. 3. Bone Transplantation--methods. 4. Osteotomy--methods. 5. Surgical Flaps. WU 600 O85 2009] RK667.I45O93 2009 617.6’93--dc22 2009043444
© 2010 Quintessence Publishing Co Inc
Quintessence Publishing Co, Inc 4350 Chandler Drive Hanover Park, IL 60133 www.quintpub.com
All rights reserved. This book or any part thereof may not be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, or otherwise, without prior written permission of the publisher.
Editors: Bryn Goates and Lisa C. Bywaters Cover and internal design: Gina Ruffolo Production: Sue Robinson Illustrations: Peter Jurek
Printed in China
Table of Contents
Dedication
The Le Fort I osteotomy, as developed by Dr William Bell, sparked a worldwide revolution as it became perfected by Dr Bell and his many colleagues. From the Le Fort I came not only bigger, more expansive operations but also smaller segmental osteotomies. Now it has led to the subject of this book, even smaller fragmentary procedures called osteoperiosteal flaps, which are flaps of edentulous alveolar bone used in reconstruction with dental implants.
In one seminal publication, Dr Bell observed that engorged collateral vascularization in the palate and labial gingival periosteum in monkeys developed immediately or soon after Le Fort I procedures. In later healing phases, a massive vascular proliferative response developed without significant scarification of the osteotomy site. Osseous gaps healed via well-vascularized bone modeling. Concern about avascular necrosis and non-union as intrinsic outcomes of a Le Fort procedure was forever put to rest. The bone was alive and nourished by the vascular pedicle even during the surgical procedure.
This famous innovator, a man who systematically has addressed maxillary orthognathic surgery from the biologic, functional, and esthetic standpoints, must be credited, as much as anyone, for this idea that a pedicled vascularized bone can be moved and subsequently heal into a new place. Thus, it is to Dr William Bell that I dedicate this book, with deference, humility, and respect.
Contributors
Michael Alterman, DDS Resident in training Department of Oral and Maxillofacial Surgery The Hebrew University of Jerusalem Jerusalem, Israel
Jean-Francois Bedard, DMD, Cert Prosthodontist Private Practice Prosthodontics Englewood, Colorado
William H. Bell, DDS Clinical Professor Department of Surgery University of Texas Southwestern Medical Center Dallas, Texas
Joel Berger, DDS, MD Private Practice Oral and Maxillofacial Surgery San Diego, California
Philip J. Boyne,* DMD, MS Professor Emeritus Department of Oral and Maxillofacial Surgery Loma Linda University Loma Linda, California
Nardy Casap, DMD, MD Professor Department of Oral and Maxillofacial Surgery The Hebrew University of Jerusalem Jerusalem, Israel
David L. Cochran, DDS, PhD Professor and Chairman Department of Periodontics The University of Texas Health Science Center at San Antonio San Antonio, Texas
Fabio Costa, MD Department of Maxillofacial Surgery University of Udine Udine, Italy
Jared Cottam, DDS, MD Private Practice Oral and Maxillofacial Surgery Greenwood Village, Colorado
Edward Ellis III, DDS, MS Professor Department of Oral and Maxillofacial Surgery University of Texas Southwestern Medical Center Dallas, Texas
Rolf Ewers, DMD, MD, PhD Chairman University Hospital of Cranio- Maxillofacial and Oral Surgery Medical University of Vienna Vienna, Austria
Benjamin Foley, DDS Resident in training Department of Oral and Maxillofacial Surgery Vanderbilt University Nashville, Tennessee
Ghali E. Ghali, DDS, MD, FACS Head Department of Oral and Maxillofacial Surgery Louisiana State University Health Sciences Center at Shreveport Shreveport, Louisiana
Paul Glick, DDS, MS Private Practice Periodontics and Implant Surgery Aurora, Colorado
Alan S. Herford, DDS, MS Chairman and Program Director Department of Oral and Maxillofacial Surgery Loma Linda University Loma Linda, California
Hideharu Hibi, DDS, PhD Associate Professor Program in Cell Information Medicine Nagoya University Nagoya, Japan
Masaki J. Honda, DDS, PhD Department of Oral and Maxillofacial Surgery Nagoya University Nagoya, Japan
Ole T. Jensen, DDS, MS Private Practice Oral and Maxillofacial Surgery Greenwood Village, Colorado
Robert Kammer, DDS Private Practice Orofacial Pain and Restorative Dentistry Boulder, Colorado
K. Lee Kuhlke, DDS, MS Private Practice Prosthodontics Englewood, Colorado
Zvi Laster, DMD Chairman Department of Oral and Maxillofacial Surgery Poriya Hospital
Tiberias, Israel Aldo Leopardi, BDS, DDS, MS Private Practice Prosthodontics Greenwood Village, Colorado
Dan Llop, CDT President and CEO nSequence Center for Advanced Dentistry Reno, Nevada
Robert Mogyoros, DMD Private Practice Oral and Maxillofacial Surgery Philadelphia, Pennsylvania
Adi Rachmiel, DMD, PhD Deputy Director Department of Oral and Maxillofacial Surgery Rambam Medical Center Haifa, Israel
Massimo Robiony, MD Department of Maxillofacial Surgery University of Udine Udine, Italy
Yoshinori Shinohara, DDS, PhD Section of Fixed Prosthodontics Kyushu University Fukuoka, Japan
Yoshinori Sumita, DDS, PhD Department of Oral and Maxillofacial Surgery Nagoya University Nagoya, Japan
Boyd Tomasetti, DMD, MS Private Practice Oral and Maxillofacial Surgery Littleton, Colorado
Shuhei Tsuchiya, DDS, PhD Division of Stem Cell Engineering Institute of Medical Science University of Tokyo Tokyo, Japan
Minoru Ueda, DDS, PhD Professor Program in Cell Information Medicine Nagoya University Nagoya, Japan
Yoichi Yamada, DDS, PhD Center for Genetic and Regenerative Medicine Nagoya University Nagoya, Japan
Preface
Why another book on alveolar bone graft augmentation? Because it is time to abandon what works. The paradox of scientific progress is that, to advance a discipline, clinicians must from time to time rethink a problem that they believe has already been solved. Such is the case with alveolar bone grafting. We must now move away from the idea of implant success to one of gingiva-alveolus-implant success, a much different goal than simple implant longevity based on a life table.
If this idea is true, then we must immediately rewrite the history of implant dentistry because historical significance changes to one of alveolar reconstruction and not simply insertion of titanium into bone. The surgical discipline of alveolar reconstruction really began with manipulation of alveolar bone and not implantation of metallic devices. Under this interpretation, the seminal developments included not only Maggiolo’s first report of the use of an endosseous implant for a dental restoration, in 1809, but Simon Hullihen’s first use of an alveolar osteotomy to reposition jawbone structure in 1849.
The latter discovery seems to be of forgotten importance for establishing dentoalveolar function in implant dentistry. If this is so, we have come back around to meet ourselves at yet another new beginning, that is, to perform jawbone osteotomies for alveolar reconstruction, after having spent the last three decades considering dental implant position as primacy for success in dentoalveolar restoration.
Discussion of the use of alveolar osteotomy for dental implantology has been remarkably absent in the literature, which has been instead dominated by studies of guided bone regeneration and/or various techniques for block bone grafting, which have been shown to be effective enough to get us by. So must we abandon what works?
There is always a sense of cognitive dissonance that arises when someone suggests that further scientific advancement is possible in a field you know well, in this case alveolar bone grafting. However, critical appraisal of bone graft performance is lacking in the literature. The success of current alveolar and sinus grafting procedures is notoriously measured by implant osseointegration or implant retention, often in settings where the implants are not relying on those bone grafts in order to succeed.
This book makes the argument that alveolar bone should have greater sway in determining the success of an implant restoration. Restoration involves not just a tooth or a root form but alveolar morphology and gingival soft tissue. Ideally the alveolar bone must be restored to its desired form, function, and vitality in what has been termed orthoalveolar form. Soft tissue generally follows suit, and implant placement then follows, creating a functional gingiva-alveolus-implant matrix.
With the use of osteoperiosteal flaps, the surgeon manipulates available bone to recover what is missing in a very special way: endosteally. Because of this relatively closed wound approach, it is as if the epigenetic signal, designed long before we wondered how a deficient bone could be improved, is suddenly turned on within the gingivoalveolar complex and the augmentation develops analogous to primordial growth.
The various osteoperiosteal flaps discussed herein are procedures that not only can be easily learned and result in fewer complications but also probably lead to a more vital alveolar reconstruction. Once mastered, bone flaps can almost entirely eliminate the need for block grafting or guided bone regeneration.
What then are the advantages and disadvantages of the osteoperiosteal flap?
Advantages:
Resulting changes in the alveolar crest are stable.Gingival architecture is maintained.The transported segment is vascularized.The periosteum is left intact and a relatively closed flap is used.The procedure results in early periosteal and endosteal osteogenesis.The operating time is relatively brief.Infection and dehiscence rates are low.Disadvantages:
It is a technical procedure with relatively blind operator access.Sufficient bone mass is needed to allow use of the osteoperiosteal flap.The vector of segment movement can be unfavorable.Vertical movement is often limited to 5 mm.For dental implant reconstruction, I have used various bone flaps for the past 20 years and am using guided bone regeneration and block grafting less and less each year. These historically important procedures are now largely absent from my surgical protocol.
For example, the book flap, although only recently described as a technical note, has been used for about 15 years with little or no complication for sites with width deficiencies of 2 mm or more. For a single-tooth site, the book flap procedure generally takes about 30 minutes to perform, and the patient generally experiences very little postoperative pain or morbidity. The dimensional stability of the augmentation is excellent and wound breakdown is negligible. In short, in a busy practice, the book flap is an easy way for both the patient and the surgeon to achieve the desired goal. Such is the case for the entire osteoperiosteal flap repertoire compared to alternative therapies.
This text is written and edited for the “wet finger” clinician—the private practice clinician, who must use techniques that work consistently, have minimal morbidity, and are simple and relatively quick. The osteoperiosteal flap, shown here in all its permutations, fulfills these criteria beautifully.
Acknowledgments
I would like to acknowledge my wife Marty, my children Sverre, Autumn, and Trygve, and my grandchildren as the underlying inspiration and drive for any success I might have outside the home.
I especially want to thank those who contributed so greatly to making this book possible: my publisher Tomoko Tsuchiya for taking a chance on me once again, Lisa Bywaters for her extreme patience and expert guidance, Bryn Goates for her positive and concise editing, Sue Robinson on an artistic layout and design, and Peter Jurek for his outstanding renderings.
I also want to express special thanks to Karen Shoop, my implant coordinator and brain away from home, Kristen Stifflear, who gave birth to a child this year and still provided many of the photos and essential content organization for the book, and my fantastic surgical assistants Cindy Formanek and Jennifer Patrick.
Dr Jared Cottam contributed as a research assistant; special thanks to him. Without my staff and associates, this decade-long process could not have been completed.
Introduction
A Curious Surgeon’s Role in the Evolution of Orthognathic Surgery in America
William H. Bell, DDS
The sine qua non of successful tissue transplantation is preservation of viability.
—William Bell
When I finished my oral surgery training 55 years ago in Houston, Texas, only a handful of surgical procedures had been developed to treat patients with dentofacial deformities. Most corrections were accomplished extraorally in the mandible. Orthodontists had virtually no interest in surgery except for the treatment of mandibular prognathism, and surgeons had very little interest in orthodontics. With only empirically based surgical techniques available, all done without efficient orthodontics, and virtually no surgery in the maxilla, it is not surprising that most patients received compromised, unesthetic, and inefficient treatment for their dentofacial deformities.
The present state of the art and science includes many more surgical procedures, most of which are performed intraorally; an improved understanding of the biologic and surgical principles; routine orthodontic treatment; and worldwide interest in the correction of dentofacial deformities. Present day surgical-orthodontic techniques are the harvest of yesterday’s research. An understanding of the evolution of these procedures is key to continued progress in this field.
A Look Back
In 1954, I was an oral surgery intern in New York City at the Metropolitan City Hospital. In any given week we saw literally hundreds of patients who were candidates for either orthognathic surgery or orthodontics. Unfortunately none of them received either treatment.
In 1955, I was an oral surgery resident at Jefferson Davis Hospital in Houston, Texas. This program was reputedly one of the nation’s finest programs in reconstructive mandibular surgery. Edward C. Hinds, the program director, was developing his innovative extraoral vertical ramus osteotomy technique for correction of mandibular prognathism. He treated many of these cases and other challenging deformities, all without maxillary surgery. I know because I was his senior resident. There was no shortage of mandibular trauma patients; midfa-cial fractures were occasionally managed by the oral surgery service.
In 1956, I became the first oral surgery teaching fellow at MD Anderson Hospital in Houston, Texas. My surgical experience at the country’s premier cancer hospital was limited to extracting teeth for two world-renowned head and neck cancer surgeons, William McComb and A. J. Ballantyne. They occasionally delegated me to treat a benign tumor or cyst of the jaws.
My curiosity and interest were profoundly influenced by observation of the cancer surgery performed by these two giants in the head and neck specialty. Patients were frequently treated by radical neck dissections and concomitant tracheotomy, with or without radiation, and maintained in the hospital for weeks without reconstructive surgery. The patient’s quality of life after such surgery was dismal and the prognosis was generally poor.
Initial Animal Studies
During my fellowship year and the following 6 years, I assisted Dr Hinds in the clinical programs at the University of Texas Dental Branch in Houston, Texas. Technical descriptions of anterior and posterior maxillary osteotomies in the literature elicited great interest but virtually no clinical application. My initial response to these procedures was not only an insatiable curiosity but also fear of the unknown clinical consequences. This same fear dominated the thoughts of virtually everyone in the oral surgery and basic science departments.
During my fellowship, I searched the available literature for relevant studies but found none that suggested a biologic foundation for these procedures. Having no research training or background, I sought the help of other colleagues. Sumpter Arnim, a fascinating and inquisitive individual, supported my curiosity with great enthusiasm and described the benefits of research for the specialty and patients. He reinforced his position with an offer of six large, white rabbits that I could use for a pilot study.
The race to find some type of research methodology continued. The goal was to demonstrate the biology of wound healing after maxillary osteotomy and the effect that maxillary osteotomies had on the viability of the dental pulp and bone. The revascularization and histologic techniques described by Rhinelander and Baragry,1 two orthopedic surgeons, seemed promising. A visit to their laboratory in Cleveland, Ohio, to observe their ongoing orthopedic revascularization studies was an eyeopening experience.
On returning to Houston, we carried out preliminary pilot studies in two of the rabbits to establish the details of microangiographic and histologic laboratory techniques. Two experimental study animals were killed 1 and 3 weeks after anterior maxillary osteotomy and compared with an unoperated control animal. The angiographic studies demonstrated that the surgical sites were amazingly well vascularized compared to the sites in the control, untreated animal—as if no surgery had been done. Hypervascularity of the surgical sites without evidence of ischemia was noted within a week in the experimental animals (Bell, unpublished data, 1963).
Because we believed that these surprising results must be mistaken, surgery was performed on two more animals. Microangiograms taken 1 and 3 weeks after surgery confirmed the initial observations. The results gave support to similar canine studies in which the surgical and laboratory techniques were refined (Bell, unpublished data, 1963).
After celebrating this success, we received National Institutes of Health grant support for comprehensive wound healing studies in primates. One-stage and two-stage anterior maxillary and posterior maxillary osteotomies were initially performed via various flap designs to determine the validity of vascularity to the repositioned osseous segments.2,3 The animals were killed 1, 3, 6, and 24 weeks after surgery for microangiographic and histologic investigation. Serial 1-mm transverse, sagittal, and horizontal tissue slices were cut from the specimens for microangiographic study. Each 1-mm tissue slice was then cut into seven microscopic slices for histologic study.
Microangiographic and histologic techniques demonstrated that intraosseous and intrapulpal circulation to the anterior maxillary segment was maintained when soft tissue was kept intact. Osteonecrosis was minimal and vascular ischemia was only transient when the anterior maxillary bone segment was pedicled to the labiobuccal mucoperiosteum, palatal mucoperiosteum, or a combination of both. These studies are described in more detail in chapter 1.
First Clinical Application
The encouraging findings after anterior and posterior maxillary osteotomies in rabbits, dogs, and monkeys suggested that similar procedures involving one or two dentoalveolar segments might be successful in human patients. The repositioning of these small dentoalveolar segments presented many imaginative and challenging possibilities. The positive results of segmental surgical procedures in primates seemed to indicate that similar results would be achieved by repositioning of small dentoalveolar segments in humans.
Pat Allesandra, a friend and colleague who specialized in orthodontics, presented at my office with a patient who displayed a 5-mm maxillary interincisal diastema. For esthetic reasons and because of the instability of such orthodontic procedures at the time, the patient sought immediate correction of the large diastema. Dr Allesandra had recently heard me discuss the results of animal studies and saw no reason why the procedure could not be done successfully in humans. Within 2 weeks, the patient was scheduled for an in-office surgical procedure.
A labial and buccal mucoperiosteal flap was raised from the underlying bone to facilitate immediate closure of the diastema by ostectomy between the central incisors and interdental osteotomies in the canine–lateral incisor interspaces.2,4 After subapical osteotomy to connect the vertical bone cuts, the dento-osseous segments were immediately repositioned to close the space. Following stabilization of the segments with stainless steel wire ligatures and brackets bonded to the teeth, the postoperative follow-up was uneventful. A long-term follow-up revealed minimal interseptal or crestal alveolar bone loss in the osteotomy and ostectomy sites. The teeth have tested vital over this period.
It seemed obvious that subsequent surgeries to reposition large dento-osseous segments should be possible if the surgical procedures involved similar wound healing principles that maintain pedicled dento-osseous segments. Results in this first case, and many similar cases, were biologically founded on the results of primate studies. The results generated enormous confidence in the possibility of surgical repositioning of one- and two-tooth dentoalveolar segments in the maxilla and mandible. Healing of interdental osteotomy sites without root resorption has since been routinely observed.
This initial surgery was performed through a circum-sulcular incision. More recently, however, physiologic split gingival incisions or circumvestibular incisions have been used. In a few of the early cases, patients were treated in two stages. In recent years, however, virtually all surgeries have been performed in a single stage in which the bone segment was pedicled to the labial buccal soft tissue flaps with minimal discernible hard or soft tissue injury.
Origin of a Successful Training Program
On June 10, 1966, at a meeting of the Houston Society of Oral Surgeons, Ashley Sills suggested that the University of Texas Dental Branch and the Houston Society of Oral Surgeons jointly sponsor an oral surgery meeting program suitable for individuals whose practices were limited to oral surgery. This was the beginning of the Edward C. Hinds Symposium, as it is known today. The first meeting was held August 5 and 6, 1966, and was attended by 140 orthodontists and surgeons.5,6 Two renowned clinicians, Alex Mohnac (oral surgeon) and Haskell Gruber (orthodontist), were the presenters. The meeting, which focused on surgical orthodontics, is still considered one of the most successful ever presented in Houston.6 At the closing reception, Dr Mohnac and I discussed the biology of maxillary osteotomy wound healing. A comment made by Dr Mohnac still reverberates to this day: “The maxilla is enveloped by viable vascularized tissue that may preclude the need of maintaining a soft tissue pedicle to the surgically repositioned maxilla because it rapidly reattaches and revascularizes to the underlying bone.”
That statement turned a light on for me. Within 2 weeks another rhesus monkey was scheduled for surgery to test this hypothesis with a nonpedicled anterior maxillary osteotomy. The postoperative result was a disaster. Within a week the surgical wound became infected, the bone was necrotic, and the maxillary teeth became nonvital and exfoliated. That particular study was terminated. Dr Mohnac’s statement, however, has forever influenced my clinical judgment whenever I am tempted to reposition a dento-osseous segment with a questionable soft tissue pedicle. It solidified my confidence that maintenance of an attached soft tissue pedicle to a repositioned maxillary dento-osseous segment is necessary to assure viability of the repositioned maxillary teeth and bone.
Move to Dallas
During 6 years of work at the Dental Science Institute in Dallas with Bernard Levy and colleagues, I had the opportunity of learning research methodology and applying the wound healing principles learned from animal investigations to clinical challenges by performing segmental surgery via osteoperiosteal flaps.
In 1971, I was recruited by Robert Walker at the University of Texas Southwestern Medical Center in Dallas, Texas, to develop a research laboratory that focused on the blood supply and wound healing associated with maxillary and mandibular osteotomies. The position offered the opportunity and environment to continue animal and clinical investigations and to apply the knowledge to solving clinical challenges and establishing a biologic foundation for repositioning the maxilla.
For the next 20 years, I had the privilege of working with and learning from creative and talented oral and maxillofacial surgery residents in the Oral and Maxillofacial Surgery Research Laboratory at the University of Texas Southwestern Medical School and the Parklandprogram. Each of these individuals made significant and unique contributions to the evolving field of orthognathic surgery, and a high proportion of these individuals have remained in teaching institutions.
Dr Walker held court at the exceptional Wednesday night conferences. All residents, fellows, and staff attended this centerpiece conference for learning. Doug Sinn, Steve Hill, Kevin McBride, Joe Kennedy, Tim Turvey, Ray Fonseca, Cesar Guerrero, and Larry Wolford all made vital contributions to this atmosphere of learning, not to mention Richard Finn, the lauded voice of craniomaxillofacial anatomy for 25 years. (Through Dr Finn’s efforts as Chief of oral and maxillofacial surgery at the Veterans Administration Hospital in Dallas, a resident position in that hospital has been one of the most sought after in the country.) Dentofacial deformity conferences were my responsibility to organize. Bruce Epker added his exceptional diagnostic and treatment planning skills for craniomaxillofacial deformities at these weekly conferences. Joe Jacobs entered into the mix at the dentofacial deformity conferences with his always sound orthodontic treatment planning skills and advice: “Do not begin treatment without a plan.” Harry Legan’s orthognathic surgery analysis contributed to the success of the orthodontic planning for the dentofacial deformity conferences.
Bruce Epker and Chuck Fish presented the Fort Worth approach to surgical orthodontic treatment planning, which frequently differed from the Parkland approach. Fort Worth promoted their “surgery first” approach and compared it with the Parkland “orthodontics first” approach. The differences in the Parkland approach of repositioning the maxilla by the Le Fort I downfracture technique and the Fort Worth approach of combined anterior and posterior maxillary osteotomies7 prompted many intense debates at dentofacial deformity conferences. The rationales for the use of the modified sagittal split ramus osteotomies (the Fort Worth way) and intraoral vertical ramus osteotomies (the Parkland way) for treatment of mandibular prognathism and selected temporomandibular joint dysfunction were keenly debated issues.
Occasional visits from my good friend and colleague in orthodontics, Tom Creekmore, provided a profound understanding of vertical dysplasias (skeletal open bite deformities) and the possible use of intraoral skeletal anchorage to treat these problems.8
Colleagues from China and Japan—Zhiahou You, Hiroshi Kawamura, Xien Chang, Jaime Quejada, Yoshinuro Yamaguchi, Toshi Hagawara—were vital to our primate studies and have since made enormous contributions to the art and science of orthognathic surgery. Chawket Mannai was instrumental in the work and publication of essential articles relative to three-dimensional repositioning of the jaws. Stephen Schendel, Heidi Ob-debeeck, and Joseph Schoeners published the first computer-based analyses of long and short face syndromes and forever influenced planning and treatment of these dentofacial deformities.
A wonderful mixture of different philosophies of surgical and orthodontic treatment, opinions, and diverse techniques practiced in the two different institutions made teaching and learning exciting and challenging for both residents and faculty. The faculty and residents served as excellent models for one another and contributed enormously to residents’ education. To this day, I maintain that the orthognathic surgical “safaris” initiated in 1973 were the most powerful and effective means we employed to help residents and visitors from every corner of the world learn the surgical, orthodontic, and biologic principles of orthognathic surgery.
Spreading the Word
The impetus to write a book at that time arose from the need for all individuals involved in treating patients with dentofacial deformity to plan, work, and treat these patients together. My typical day would include orthognathic surgery in the Parkland Hospital operating room, where numerous intraoperative photographs were taken and destined to end up at the office of the medical illustrator. In the medical illustration department, I had continued support from one of the country’s premier medical illustrators, Bill Winn. Most of these procedures had never been anatomically illustrated in detail. This was an enormous undertaking based on photographs taken during surgery, anatomy texts, and Bill Winn’s fantastic skill. His skill was a vital part of the equation because this was the precomputer age, and the illustrations were all done as original artwork.
From 1973 to 1985, the multivolume Surgical Correction of Dentofacial Deformities, the work of many talented surgeons and orthodontists, was in the making.9–11 If optimum function, esthetics, and stability were to be achieved in most patients who had a deformity, collaboration between specialists in medicine and dentistry was mandatory. The objective was to write a comprehensive clinical reference on the interdisciplinary art and science of correcting dentofacial deformities by surgery and orthodontics. This work combined an atlas of surgical and orthodontic procedures with sound diagnostic and biologic guidelines for their application.
This textbook and others by prominent researchers who had made significant contributions to the evolving discipline of orthognathic surgery helped to establish the legitimacy of the field and demonstrated this fundamental truth: When orthognathic surgical procedures are planned and executed in concert with orthodontics, virtually all dentofacial deformities can be treated effectively.
Conclusion
Fifty-five years after my experiences as an oral surgery resident at MD Anderson Hospital, I had the privilege of participating in an amazing symposium, Technological Advances in Head and Neck Oncology and Cranio-Maxillofacial Surgery. Patients are now diagnosed and treatment planned with the use of three-dimensional virtual imaging, treated surgically with microvascular reconstruction performed immediately at the time of ablation, and generally discharged from the hospital within a few days. Oral and maxillofacial surgeons with fellowship training in head and neck oncologic surgery as well as microvascular surgery perform the procedures and manage the patients throughout their cancer treatment. Many of these revolutionary techniques have evolved from the science and techniques of orthognathic and maxillofacial surgery.
The expanded role of oral and maxillofacial surgeons in the treatment of cancer patients is just one example of the way in which research in our field has benefited patients. Those who question the role of our specialty in oncologic and reconstructive surgery need to be reminded of the paradigm shift in head and neck reconstructive surgery since I was an oral surgery resident. Fifty-five years ago, I was known as an oral surgeon; today through the efforts, support, and teaching of many colleagues, I am known as an oral and maxillofacial surgeon.
Our specialty is poised to move forward to new frontiers. Many questions concerning the future of research and treatment of patients with dentofacial deformities remain unanswered. It is hoped that this book will not only provide answers to clinical questions but also stimulate further research and progress in treatment methods based on interdisciplinary animal and clinical investigations, achieved collaboratively by basic and clinical scientists.
References
Rhinelander FW, Baragry RA. Microangiography in bone healing. 1. Undisplaced closed fractures. J Bone Joint Surg 1962; 44A:1273-1298.Bell W H. Revascularization and bone healing after anterior maxillary osteotomy: A study using adult rhesus monkeys. J Oral Surg 1969;27:249–255.Bell WH, Levy BM. Revascularization and bone regeneration following anterior mandibular osteotomy. J Oral Surg 1970;28: 196–203.Bell WH. Surgical-orthodontic treatment of interincisal di-astemas. Am J Orthod 57:158, February. 1970;57:158–163.Mohnac AM. Surgical correction of maxillomandibular deformities. J Oral Surg 1965;23:393–407.Debes RR. Looking Back: A History of the Oral and Maxillofacial Surgery Training Program, University of Texas–Houston, 1949–1999. Houston: University of Texas–Houston Health Science Center University Press, 1999.Epker BN, Wolford LM. Dentofacial Deformities: Surgical-Orthodontic Correction. St Louis: Mosby, 1980.Creekmore TD, Eklund MK. The possibility of skeletal anchorage. J Clin Orthod 1983;17:266–269.Bell WH, Proffit WR, White RP (eds). Surgical Correction of Dentofacial Deformities, vol 1. Philadelphia: Saunders, 1980.Bell WH, Proffit WR, White RP (eds). Surgical Correction of Dentofacial Deformities, vol 2. Philadelphia: Saunders, 1980.Bell WH (ed). Surgical Correction of Dentofacial Deformities—New Concepts, vol 3. Philadelphia: Saunders, 1985.SECTION I
Biologic Rationale
CHAPTER 1
Biologic Basis of the Osteoperiosteal Flap
William H. Bell, DDS
To see what is in front of one’s nose requires constant struggle.
—George Orwell
In the early evolutionary years of oral and maxillofacial surgery, pulpal response to alveolar osteotomies was a central question to be answered. Relatively few surgeons, however, were interested in this fundamental question. At the annual association meetings of the American Association of Oral and Maxillofacial Surgeons or the International Association for Dental Research, it was not unusual to see only five or six surgeons in attendance in the research sessions debating the question of what constitutes a viable tooth.
It had been recognized for some time that teeth contained within a repositioned dento-osseous segment did not respond positively to electrical stimulation immediately after surgery. This aberrant testing was usually transient and results returned to normal after 3 to 8 months. A small, dedicated cadre of investigators1–6 often debated as to whether pulpal vascularity were more important than neuronal continuity.
In time, preservation of pulpal circulation was generally considered to be necessary if normal pulpal anatomy were to be preserved following dentoalveolar surgery. Neuronal, blood flow, and histologic studies gradually confirmed these findings and created enormous interest in the surgical repositioning of all maxillary and mandibular teeth by dentoalveolar surgery and orthodontics. These studies opened the gate to the possibility of simultaneous repositioning of all or a part of the maxilla and maxillary teeth independently as small dento-osseous segments.
Recent studies have used laser Doppler flowmetry to assess tooth vitality after Le Fort I osteotomy.7,8 These studies have clearly demonstrated that teeth without normal innervation can have an intact blood supply and be vital.
Fig 1-1a Anterior maxillary osteotomy performed after reflection of the labial and buccal mucoperiosteum. (Reprinted from Bell et al23 with permission.)
Biology of Wound Healing
Anterior maxillary osteotomy
Maxillary deformities have been recognized and described for centuries, but the challenge to correct them through surgery in the anterior maxilla was not met until the turn of the century. Bold attempts to move the anterior maxilla were first made by Cohn-Stock,9 Wassmund,10 and Spanier,11 who were unaware of the biologic basis for the healing of such surgically created wounds. Analysis of Cohn-Stock’s initial attempt to retroposition the anterior maxilla surgically indicates that he feared the consequences of such a procedure and attempted to avoid them by creating a greenstick fracture of the anterior maxilla through a transverse palatal incision; the retropo-sitioned maxilla subsequently relapsed.
When maxillary surgical procedures were introduced to the United States,12–15 the rationale for use of the various surgical techniques for correcting dentofacial deformities was empirical.16 Basic questions concerning the healing of surgical wounds produced by maxillary osteotomies had not been investigated. Many surgeons believed that the maxilla healed by fibrous union. Others believed absolute stability was necessary. Devitalization of teeth and bone in the mobilized segments had been re-ported. Varying degrees of relapse subsequent to posterior maxillary osteotomy9 and total maxillary osteotomy were reported. The possibility that the maxilla could be successfully repositioned superiorly or inferiorly through surgery was doubted by many clinicians and scientists. The blood vessels necessary to maintain circulation to the mobilized bony segments and teeth had not been studied. Consequently, both one-stage and two-stage procedures (of empirical duration ranging between 2 and 8 weeks) were devised to prevent impairment of the vascular supply to the mobilized dentoalveolar segments.17
Fig 1-1b Midpalatal sagittal incision for palatal osteotomies. (Reprinted from Bell et al23 with permission.)
In 1962, animal and clinical investigations were initiated to delineate the biology of maxillary osteotomy wound healing. Since then, rabbits, dogs, monkeys, and baboons have been used as experimental models to investigate the revascularization and bone healing associated with various maxillary techniques.12,16,18,19Macaca mulatta was usually selected as the experimental animal of choice because of its anatomic, physiologic, and dental similarities to the human. Because maxillary osteotomies are usually performed in adults, large male rhesus monkeys from 8 to 14 years of age and weighing an average of 9 kg, were chosen for study.
From 1962 to 1965, revascularization and bone healing were studied on animal models after clinical simulations of three variations of anterior maxillary osteotomy techniques10,14,20 (Fig 1-1) were performed via various flap designs to validate vascularity to the repositioned osseous segments.12,13,22 The animals were killed 1, 3, 6, and 24 weeks after surgery for microangiographic and histologic investigation.
Fig 1-2 Horizontal microangiogram demonstrating the vascular pattern of a control animal. A reticulated network of periodontal plexus encircles each tooth, composed of anastomosing blood vessels from the labial (facial), gingival, intra-alveolar, and apical vessels. (Reprinted from Bell et al21 with permission.)
Serial 1-mm transverse, sagittal, and horizontal tissue slices were cut from the specimens for microangiographic study, which were in turn cut into seven microscopic slices for histologic study. Microangiographic and histologic techniques demonstrated that intraosseous and intrapulpal circulation to the anterior maxillary segment was maintained when soft tissue was kept intact.12,22 Osteonecrosis was minimal and vascular ischemia was only transient when the anterior maxillary bone segment was pedicled to the labiobuccal mucoperiosteum, palatal mucoperiosteum (Figs 1-2 and 1-3), or a combination of both. Osseous union between most of the sectioned segments occurred within 6 weeks without immobilization of the mandible.
Circulation to the dental pulp was maintained when the bone cuts were made away from the apices of the teeth (5 mm when feasible, which was thought intuitively to be “safe”). In some of the early animals, before we became familiar with the anatomy of the monkey (very long, curvilinear canines), we inadvertently sectioned some of the tooth-root apices.20,23,24 When this occurred, pulpal circulation terminated and pulpal necrosis was observed.
Six weeks after the osteotomies, there was no detectable intraosseous or intrapulpal ischemia. The repar-ative response in the endosteal vascular bed appeared more intense than it did in the periosteal vascular bed. Histologically, the proximal and distal bony segments were united with cancellous bone.
Fig 1-3 Microangiogram of the premolar region of an experimental animal, 6 weeks after single-tooth repositioning, demonstrating the generalized distribution of barium sulfate in the soft tissues, bone, and pulp canals of the tooth. (Reprinted from Bell et al21 with permission.)
Twenty-four weeks after maxillary osteotomies, the periosteal and endosteal circulatory beds had been virtually reconstituted to their normal vascular architecture. The endosteal-periosteal anastomosis through cortical bone had been restored. Histologic examination of the osteotomy wounds revealed healing of the cortical bone and remodeling of the spongiosa.
Each of the three different single-stage anterior maxillary osteotomy techniques maintained blood supply to the bone and soft tissue. When the labial mucoperiosteum was completely reflected from the anterior alveolar region, collateral circulation from the palatal vessels was sufficient to replace the interrupted circulation (Fig 1-4a). When anterior maxillary osteotomy was performed through a palatal mucosal flap in combination with buccal vertical flaps in the premolar regions, the anterior maxillary bone fragment and teeth received their blood supply from an intact labial pedicle through the vascular plexuses of the gingiva and the nasal floor (Fig 1-4b).
The possibility that the anterior maxillary bone segment was a free dental osseous segment that rapidly revascularized was not supported by these studies. When the mucoperiosteum was completely reflected from the bone, intraosseous necrosis, gross vascular ischemia, and nonunion resulted.25 Nonpedicled, free anterior maxillary dental osseous segments did not revascularize, became necrotic within a week, and did not heal with the proximal bone fragment. It is clear that the viability of transposed dental osseous segments was preserved through continuous circulation supplied by attached mucoperiosteum.
Fig 1-4a Incisions of soft tissue and bone for correction of anterior vertical maxillary excess via the Cupar technique of anterior maxillary osteotomy. (Reprinted from Bell et al21 with permission.)
Some of the clinical techniques for anterior maxillary osteotomy are difficult because the palatal bone cuts are made blindly. A transverse palatal incision26,27 affords excellent access to the hard palate for this operation14 but severs palatal blood vessels. The operation may alter the circulation to both bone and teeth and reduce the viability of the mobilized segment.14
Overall, these findings suggested that analogous clinical pedicled segmental surgeries should maintain vitality through use of osteoperiosteal flaps. Selection of the individualized surgical technique that will best avoid damage to the blood supply would be dependent on the clinical objective of the surgeon.
Posterior maxillary osteotomy
In 1971, Bell and Levy25 reported on the biology of wound healing in posterior maxillary osteotomies (Fig 1-5). Their microangiographic and histologic study of single-stage posterior maxillary osteotomies in adult rhesus monkeys revealed minimal osteonecrosis, transient vascular ischemia, and osseous union between most of the osteotomized segments. When the bone cuts were made away from the apices of the teeth, pulpal circulation was preserved. Within 4 weeks after the palatal surgery, the palatal mucoperiosteum was reattached to the underlying bone, as evidenced by the revascularization of the raised buccal and palatal soft tissue flaps to the underlying bone.
Fig 1-4b Incisions of soft tissue and bone for correction of anterior-posterior maxillary excess via the Wunderer technique of anterior maxillary osteotomy. (Reprinted from Bell et al21 with permission.)
The results of these clinically analogous animal studies indicated that single-stage posterior maxillary osteotomies are biologically sound. The single-stage posterior maxillary osteotomy will become one of the most commonly used surgical procedures for correction of posterior maxillary vertical hyperplasia. A single simple subapical ostectomy or osteotomy may greatly facilitate predictable and rapid superior repositioning of the posterior maxilla through compression osteogenesis or immediate repositioning. The repositioned osseous segment may include one or more dental implants28 (Fig 1-6).
Thus, wound healing in rhesus monkeys has shown that both anterior and posterior maxillary osteotomies are a biologically sound clinical procedures when the circulation to the mobilized bone segment is maintained by attached mucoperiosteum (Fig 1-7).12,13,25 The collateral circulation occurs between osseous and soft tissue elements; intraosseous collateral circulation and vascular anastomoses are found among the periodontal, gingival, floor of the nose, and palatal plexuses (Fig 1-8). Therefore, bone and soft tissue incisions can be designed and made selectively without significantly altering the blood supply to the bone or teeth in the mobilized segment.
Figs 1-5aand 1-5b Incisions of soft tissue and bone used for posterior maxillary osteotomy in experimental animals. (Reprinted from Bell et al21 with permission.)
Fig 1-5c Intraoperative view of single-stage pos- terior maxillary osteotomy.
Fig 1-6 Correction of anterior open bite by posterior maxillary osteotomy (Kufner technique). (a) The soft tissue incision is retracted inferiorly to expose the interdental osteotomy site; the superior portion of the interproximal osteotomy is accomplished. (b) The medial wall of the maxillary sinus is sectioned between the palatal roots and the nasal floor with a curved osteotome. The palatal mucosa is preserved by carefully malleting an osteotome against the surgeon’s finger positioned at the horizontal-vertical juncture of the palate. The dentoaveolar segment pedicled to palatal mucosa and buccal gingiva is downfractured. Ostectomy of the superior, medial, and posterior aspects of the segment is accomplished. (c) Repositioned posterior segment is fixed to the zygomatic buttress with a suspension wire, which is ligated to an orthodontic arch wire fixed to a stable part of maxilla. (Reprinted from Bell et al21 with permission.)
Le Fort I segmented downfracture
Successful transposition of the maxillary dento-osseous segments by Le Fort I osteotomy depends on preserving the viability of the repositioned segment by proper design of the soft tissue and bony incisions (Figs 1-9 to 1-11). The collateral circulation within the maxilla and its enveloping soft tissues and the many vascular anastomoses in the maxilla permit numerous technical modifications of the Le Fort I osteotomy.
Vascular anastomoses between the maxilla and its enveloping soft tissues are crucial in providing compensatory blood supply to dento-osseous segments after the nutrient medullary vascular system is transsected. The normal blood supply of the maxilla originates centrifugally from the alveolar medullary arterial system29–31 (see Fig 1-8). The mucoperiosteal arterial system also gives off many branches that penetrate the cortical bone and supply blood to the maxilla (Fig 1-12). The system consists not only of capillaries but also arteries and veins, which are arranged in varied configurations.32 The multiple sources of blood supply to the maxilla and the abundant vascular communications between the hard and soft tissues constitute the biologic foundation of maintaining dento-osseous viability despite transsection of the medullary blood supply after osteotomies.32
Fig 1-7a Histology showing necrosis of nonpedicled segment at 1 week. A blood clot (far right) demarcated from the proliferating mesenchymal tissue and capillaries (young granulation tissue [G]). Spicules of necrotic bone are already adjacent to bone cut (NB) (original magnification ×180).
Fig 1-8 Blood supply to the anterior maxilla. The vascular architecture, consisting of freely anastomosing gingival plexus, palatal plexus, periodontal plexus, labial artery, intra-alveolar vessels, apical vessels, and pulp vessels, permits anterior maxillary osteotomies to be performed without compromising circulation to the anterior maxilla and teeth. (Reprinted from Bell et al21 with permission.)
Fig 1-9(a) Intraoperative view of pedicled four-segment Le Fort I osteotomy in adult Rhesus monkey showing 10-mm maxillary advancement; maxilla stabilized with interosseous wires. (b) Clinically analogous pedicled downfractured four-segment Le Fort I osteotomy technique.
Fig 1-7bPedicled segmental osteotomies will quickly revascularize with osseous union occurring at 5 to 6 weeks, wheras nonpedicled osteotomy segments undergo osteonecrosis.
Fig 1-10a Microangiogram of the canine–first premolar region of an experimental animal immediately after total maxillary osteotomy, revealing ischemia (I) in the bone encasing the canine (C) and an avascular zone (A) below the buccal and nasal mucosal flaps (F). T—vascularized premolar; Pa—palate; OS—osteotomy site; EM—extravasated injection medium. (Reprinted from Bell et al21 with permission.)
Fig 1-10b Microangiogram of the molar region of experimental animal 2 days after surgery, revealing generalized distribution of the injection medium in soft tissues, bone, and pulp canals of the molar (T) as well as an avascular space (A) between superior surface of the maxilla (M) and detached nasal mucosa (NM). Pa—palate; NC—nasal cavity. (Reprinted from Bell et al21 with permission.)
Fig 1-10c Microangiogram of molar region of experimental animal 1 week after Le Fort I osteotomy demonstrates increased filling of periosteal (P) vascular beds and (E) endeosteal circulatory bed in the margins of the osteomized bone. OS—osteotomy; Pa—palate; NC—nasal cavity; M—maxilla.
Fig 1-10d Microangiogram of the molar region of an experimental animal 4 weeks after surgery, revealing reconstitution of the circulation between osteotomized segments by proliferating vessels. (arrow) osteotomy site; MS—maxillary sinus; T—vascularized pulp canal of the molar. (Reprinted from Bell et al21 with permission.)
Fig 1-10e Histologic appearance of the osteotomy site 4 weeks after surgery. Bone segments are united by viable vascularized osteophytic new bone, osteoid, and mature fibrous connective tissue (hematoxylin-eosin stain; original magnification ×130). (Reprinted from Bell et al21 with permission.)
Fig 1-11 Experimental animal. Wound dehiscence, infection, osteonecrosis, and loosening of teeth have manifested 12 days after nonpedicled total maxillary osteotomy surgery. Similar wound healing is observed in smaller nonpedicled maxillary and mandibular dento-osseous segments. (Reprinted from Bell et al21 with permission.)
In an attempt to identify the effects of soft tissue flap design, segmentation of the maxilla, stretching of the vascular pedicle during healing, and transsection of the descending palatine vessels, clinically analogous four-piece maxillary osteotomies were performed using the Le Fort I osteotomy technique in 14 adult rhesus monkeys. The revascularization and bone healing associated with the operation were studied at various time intervals by microangiographic and histologic techniques.32 Transient vascular ischemia, minimal osteonecrosis in the margins of the osteotomized segments, and small variations in the timing of osseous union of the segments were observed in the experimental animals. Within the period of postoperative study, osseous union of the bone segments was observed.
Fig 1-12 Circulation and freely anastomosing vascular plexuses of the gingiva, palate, nose, maxillary sinus, and periodontium permit the surgeon to make many technical modifications without jeopardizing the blood supply to the maxillary dentoalveolar segments.
Results of this qualitative study indicated that palatal mucosa and the labiobuccal gingival mucosa provide an adequate nutrient pedicle for single-stage Le Fort I maxillary osteotomies. Segmentation, stretching of the vascular pedicle, and transsection of the descending palatine vessels have no long-term discernible effect on revascularization or bone healing associated with the technique 13,20,30,31 (see Fig 1-10).
Preservation of the integrity of the descending palatine vessels has not been found to be essential to maintaining maxillary viability.29,32 Experimental blood flow studies have shown that the immediately postoperative volume of blood flow after Le Fort I osteotomy was much lower in the maxilla with a transsected and ligated descending palatine vessel than it was in the maxilla in which the integrity of the vessel was preserved.30,31,33 However, You and colleagues31 showed that transsection of the descending palatine vessels had no discernible effect on bone or soft tissue healing. The combination of angiography and histology provides a means of qualitatively studying the effects of surgery on the intact blood supply of the osseous and dental pulp cells.
Fig 1-13(a to c) Small dentoalveolar segments can be repositioned to move single-tooth or two-tooth sites into occlusion. (d) Freely anastomosing vascular plexuses of the gingiva, palate, periodontium, and pulp.
Surgical repositioning of single-tooth dento-osseous segments
After the development of predictable, biologically based techniques for immediate repositioning of the anterior and posterior maxillary dentoalveolar segments, which usually contained between three and six teeth, the challenge remained as to whether single-tooth dento-osseous segments could be repositioned similarly. Because pedi-cling the segment to a relatively small amount of soft tissue could presumably imperil circulation to the mobilized dento-osseous segment, many surgeons have avoided such procedures. The biology of healing associated with the surgical repositioning of single-tooth dento-osseous segments was studied in two different surgical techniques performed in adult mongrel dogs.34
The adult mongrel dog serves as an excellent experimental model for studying biologic and surgical principles involved in the immediate repositioning of single-tooth dento-osseous segments. The interdental spacing (approximately 1 to 2 mm) is comparable to many clinical situations in which immediate repositioning of small dento-osseous segments by vertical interdental and subapical osteotomies is currently considered feasible.34
Microangiographic and histologic studies of both one-stage and two-stage techniques for immediate surgical repositioning of single-tooth dento-osseous segments—analogous to segmental osteotomies of edentulous sites for dental implants—revealed early but transient vascular ischemia, minimal osteonecrosis, and osseous union between the osteotomized segments33 (Figs 1-13a to 1-13d). The attached soft tissue provided an adequate nutrient pedicle for immediate repositioning of single-tooth dento-osseous segments by interdental and subapical osteotomies. With these experimental surgeries, there were no advantages to the use of the two-stage procedure from a biologic point of view. The results supported the clinical use of techniques that maximize the attachment of the mucogingiva to the mobilized dento-osseous segment.34 Clinical trial and application of both of these techniques for immediate repositioning of single-tooth dento-osseous segments is justified.
Fig 1-13(e to g) This procedure was a forerunner to small single-tooth segmental osteotomies later treated with dental implants.
Successful transposition of dento-osseous segments depends on preservation of viability by proper design of the soft tissue and bony incisions. The collateral circulation within the maxilla and its enveloping soft tissues and the numerous vascular anastomoses in the maxilla permit many technical modifications of the two techniques used in the investigation. These early studies further add credibility to the use of small segmental osteotomy fragments of edentulous bone as long as the vascularized pedicle remains intact (Figs 1-13e to 1-13g).
Clinical Application of the Le Fort I Osteotomy
From the initial description of the downfracture technique for segmentation of the maxilla until the present time, approximately two thirds of Le Fort I osteotomies have been segmental operations.29–33 The descending palatine vessels were either intentionally or inadvertently transsected in approximately one-third of these cases, without discernible clinical consequences. In all probability, however, the vessels were unknowingly transsected in even more cases. A reasonable effort is routinely made to preserve the integrity of these vessels whenever feasible; there is, however, no reluctance to clamp them with a vascular clip or cauterize them to gain accessibility or enhance visualization. Superior or posterior repositioning of the posterior portion of the maxilla is a movement in which transsection of the vessels may most frequently be indicated in order to visualize and gain access to the tuberosity–pterygoid plate junction and to reposition the maxilla.
Variations
In principle, surgical repositioning of small dentoalveolar segments by the downfracture technique is biologically and clinically sound. In clinical practice, to improve the tooth alignment or move edentulous segments, one or multiple interdental osteotomies can be planned to move segments that are one or more teeth in length.17,34–41
The Le Fort I osteotomy may be designed to reposition small anterior and posterior maxillary dento-osseous segments simultaneously37 (Figs 1-14 to 1-18). Maximal palatal or labiobuccal soft tissue attachment must be maintained. Without proper planning and precise technique, mobilized dentoalveolar segments may become so small that any movement may devascularize and devitalize them. In each case, the design of the bony and soft tissue incisions should be individualized to maintain the largest possible dento-osseous segment to preserve the largest possible soft tissue pedicle; the larger the osseous segment, the greater the amount of palatal mucosa. In this way, a single-tooth dento-osseous segment can still retain an adequate soft tissue pedicle.
Fig 1-14a Sectioning. The anterior maxilla is partially divided into two segments before the lateral osteotomies are accomplished. (left) The mucosa is retracted to visualize the labial osteotomy site. The interdental osteotomy, incompletely accomplished with a fissure bur, extends from the anterior aspect of the nasal floor inferiorly to the crest of the alveolar ridge. (right) Superiorly, the interdental osteotomy is deepened into the spongiosa; more inferiorly, a corticotomy is made. (Reprinted from Bell and Guerrero37 with permission.)
Fig 1-14b Interincisal osteotomy is accomplished with the reciprocating saw blade placed superiorly into the piriform aperture. The skin hook is placed inferiorly to facilitate visualization and prevent injury of the attached gingiva. (Reprinted from Bell and Guerrero37 with permission.)
Fig 1-14c Sectioning. (left) A finger is positioned on the palate to feel the blade transect bone. (right) An osteotome is sequentially malleted into the interradicular area, proceeding inferosu-periorly until it partially transects the palatal bone and its tip makes contact with the nasal floor immediately lateral to the anterior nasal spine. (center) Subperiosteal tunneling dissection of the anterior maxilla, infraorbital nerve, and anterior nasal septum, and lateral nasal walls superiorly to the base of the inferior turbinate. Given that the anterior-inferior margin of the piriform rim is usually elevated above the nasal floor, care must be taken to remain in a subperiosteal plane by dissecting inferiorly and posteriorly from the inferior piriform rim. The dissection is carried to the posterior aspect of the hard palate, onto the base of the nasal septum approximately 5 mm above the nasal floor, and then to the base of the inferior turbinate on the lateral nasal wall. (Reprinted from Bell and Guerrero37 with permission.)
Fig 1-14d Mobilization. The osteotome is malleted into the interseptal area between the central incisors to fracture the crestal alveolar bone. Digital pressure on the palate indicates when the osteotome has transected the palatal cortex. This is important to prevent damage to the palatal mucoperiosteum, which is the principal blood supply. Incomplete splitting of the anterior maxilla is facilitated by malleting (1) and manipulating a sharp osteotome into the center of the stable maxilla. The two segments are made partially mobile by careful torquing (2) and lateral manipulations of an osteotome. (Reprinted from Bell and Guerrero37 with permission.)
Almost any combination of one-, two-, three-, four-, five-, or six-tooth segments may be simultaneously mobilized and selectively repositioned to achieve the desired alignment by a combination of interdental or subapical corticotomies and/or osteotomies (see Fig 1-14a). Corticotomies are used for sectioning, perforating, or mechanically altering the cortical bone to the depth of the medullary bone, which remains intact; they are used to facilitate repositioning of teeth predominantly by tipping movements.42,43 Osteotomies are for sectioning both the cortical and medullary bone to create a bone segment; interdental osteotomies may be used for selective bodily movement of dentoalveolar segments by distraction osteogenesis. 17,40–44
Fig 1-15 A high-level Le Fort I osteotomy is customized to achieve variable repositioning of the maxilla (arrows). This is frequently accomplished without bone grafting. (Reprinted from Bell and Guerrero37 with permission.)
Fig 1-16 With the maxilla downfractured, the posterior maxilla is widened by a midpalatal sagittal osteotomy accomplished with a straight reciprocating saw blade. A finger is positioned on the palate to feel the blade when it partially transects the cortical plates. The bone is then completely sectioned and mobilized with an osteotome malleted along the intended line of osteotomy. (Reprinted from Bell and Guerrero37 with permission.)
Fig 1-17a The maxilla has been repositioned laterally to create a 10-mm gap between the incisors. (Reprinted from Bell and Guerrero37 with permission.)
Fig 1-17b The segments are maintained in the planned position by a rapid palatal expander as the incisors close the distraction gap spontaneously (without active orthodontics). (Reprinted from Bell and Guerrero37 with permission.)
Simultaneous tipping and retraction of proclined and prominent maxillary anterior teeth or uprighting and advancement of retroclined anterior teeth can be accomplished by segmental Le Fort I osteotomy. Arch length can be increased by this procedure, greatly improving treatment efficiency and avoiding the need for maxillary and mandibular extractions in selected cases.
Periodontal and periapical problems associated with interdental osteotomies are minimized if the Le Fort I downfracture technique is used to gain simultaneous access to and direct visualization of labiobuccal and palatal aspects of the areas where they are planned. Meticulous and precise surgical technique, prudent selection of osteotomy sites, good lighting of the surgical field, careful use of thin sharp osteotomes, the use of ultrasonic osteotomy (piezoelectric surgery),45 with its soft tissue–sparing effect, and the use of directly bonded orthodontic appliances are vital adjuncts to safe and successful interdental and subapical osteotomies.
Fig 1-18 Surgically facilitated prosthetic and orthodontic reconstruction of the maxilla using the versatility of the Le Fort technique. Palatally inclined (Pa) teeth repositioned by interdental and subapical corticotomies. Bucally inclined (Bu) teeth repositioned by interdental and subapical osteotomies.
Fig 1-19a Histologic appearance of pulp (P) of a control maxillary third molar crown; odontoblastic layer (O) and adjacent pulpal tissues; dentin (D); and blood vessel (BV) (hematoxylin and eosin and Bodian stains; original magnification ×10). (From Di et al.50 Reprinted with permission.)
Fig 1-19c Bone-impacted maxillary third molar buds (arrows) before surgery. (From Di et al.50 Reprinted with permission.)
Segmentation of the anterior maxilla to improve the axial inclination of the anterior teeth without ostectomy or extractions may be the treatment of choice in selected cases; the desired anteroposterior position is achieved by Le Fort I osteotomy.
Potential complications
There have been relatively few reports of the loss of small or large maxillary dento-osseous segments after Le Fort I osteotomies. A study of the circumstances involved when segments are lost generally reveals that the operat-ing surgeon has violated a basic biologic or surgical principle. Most frequently, the vascular pedicle has not been maintained by proper soft tissue flap design, or circulation to the mobilized segment has not been preserved by way of attached palatal mucoperiosteum. Excessively long and traumatic surgery, imprudent selection of interdental osteotomy sites, strangulation of the circulation by improperly positioned suspension wires or imprudent use of palatal splints, poor visualization of the surgical site because of insufficient blood pressure control, and excessive stretching of the palatal mucosal pedicle are other causes of compromised wound healing.
Fig 1-19b Histologic appearance of the pulp of a maxillary third molar crown 50 months after surgery. The tooth demonstrates an intact and well-aligned odontoblastic layer (O) and normal adjacent pulpal architecture; D—dentin; N—nerve; P—dental pulp (hematoxylin and eosin and Bodian stains; original magnification ×10).
Fig 1-19d Same teeth fully developed and erupted (arrows) at follow-up, 28 months after Le Fort I surgery. (From Di et al.50 Reprinted with permission.)
Devascularization and devitalization of a repositioned segment may be caused by excessive stretching or detachment of the soft tissue pedicle or by inappropriately designed palatal soft tissue incisions. Such complications can be avoided by the use of precise three-dimensional cone-beam computed tomography studies to determine the three-dimensional positional changes of the segments, careful three-dimensional anatomic cast surgery, and/or or virtual imaging and meticulous surgical technique during the operation.





























